Fact Sheet
Constructing a Target Product Profile – Industry’s Perspective
A Target Product Profile (TPP) is a framework for the development of therapeutic candidates that summarises key features of an intended commercial product. A well-designed TPP provides a clearly articulated set of goals that help focus and guide development activities to reach the desired commercial outcome.
The aim of this document is to highlight the key considerations required for construction of a TPP from an industry perspective.
Consideration of the following is essential in construction of a high value TPP:
- Target Population (i.e., defining the specific unmet need in a subset of patients)
- Differentiation from current treatment options or standard of care, and
- Administration and Dosage considerations
An indicative template for a TPP is provided below. Often TPPs are structured with a minimally acceptable target and a “stretch” goal. Failure to meet the parameters defined as “essential” will often mean termination of product development. Exceeding these parameters or meeting the “ideal” profile will likely increase the value of the product.
Target Population
The target population will be that cohort of patients for which there is inadequate treatment, i.e., there is an unmet medical need.
In addition to precisely stating the unmet need, the following should be considered when selecting and defining the target population:
- Segmentation – Are there specific subsets of patients/diagnoses for which the program is targeted to treat?
- Epidemiology – What is the incidence and/or prevalence of the specific condition?
- Quantification of the unmet need – how is the unmet need measured? what proportion of patients are inadequately treated by this measure? Over what period?
Differentiation from Current Treatment Options or Standard of Care
The TPP will define how the program is differentiated from current treatment options or standard of care therapies. Clear measures of efficacy and/or safety should be listed. The following should be considered:
Efficacy Endpoints
- Validated efficacy endpoints used in Phase III clinical trials for the selected indication should be defined. FDA produces many guidelines and frequently defines their expectations around which endpoints should be used when seeking approval for specific indications.
- Ideally efficacy endpoints are objectively defined with clear measures of success (e.g., delta with comparator and what constitutes a clinically meaningful level of improvement?).
- Consideration should be given to what is a primary versus secondary endpoint. The time an endpoint is measured, length of follow up, relapse rates and expected performance against relevant benchmarks should be noted.
- Approved products have publicly available package inserts which contain key data of use in building a TPP (g., what endpoints were used, and the efficacy results that led to regulatory approval).
- Generally an essential endpoint measure will have at least equivalent efficacy to standard of care or first-line treatment in the target indication, while the ideal measure should exceed this level of efficacy. In addition, ideal efficacy measures may include potential secondary endpoints that are commonly used in Phase III clinical trials.
Safety Endpoints
- The required safety profile for the target product should also be defined in the TPP.
- This should demonstrate at least equivalence, or differentiation from the safety profile of current treatment options or standard of care (again package inserts of approved products in the indications being sought are useful in this regard).
- Specific adverse effects of current treatment options or standard of care should be defined (g., by incidence and severity) and the level of improvement or equivalence sought.
Administration and Dosage Considerations
How the target product will be administered should also be clearly defined in the TPP. The following should be considered:
- Will the target product be used for treatment or prevention?
- Will the target product be used as a monotherapy or in combination with other current treatments?
- What is the proposed route of administration?
- Will any titration be needed to effective dose?
- What is the proposed dosage schedule?
If the intention is to differentiate the target product from the current market through improvement of convenience of administration, then this should also be clearly defined in the TPP.
Summary
In summary, a TPP focuses research on product conceptualisation. A TPP should clearly define:
- The target population and condition, including definition of specific subsets of patients to be treated
- Efficacy endpoints and the required level of improvement to differentiate from products currently on the market
- Proposed safety profile, and
- Administration and Dosage of the target product.
Once completed, a TPP will help articulate the value proposition of a potential product (e.g., how many lives will my product save? How many patients do I need to treat to save one? How many complications will it avoid?) Consideration of the competitive landscape (e.g., cost to treat or product sales) and where this product fits in will help define the potential market share and market value.
Template for a TPP
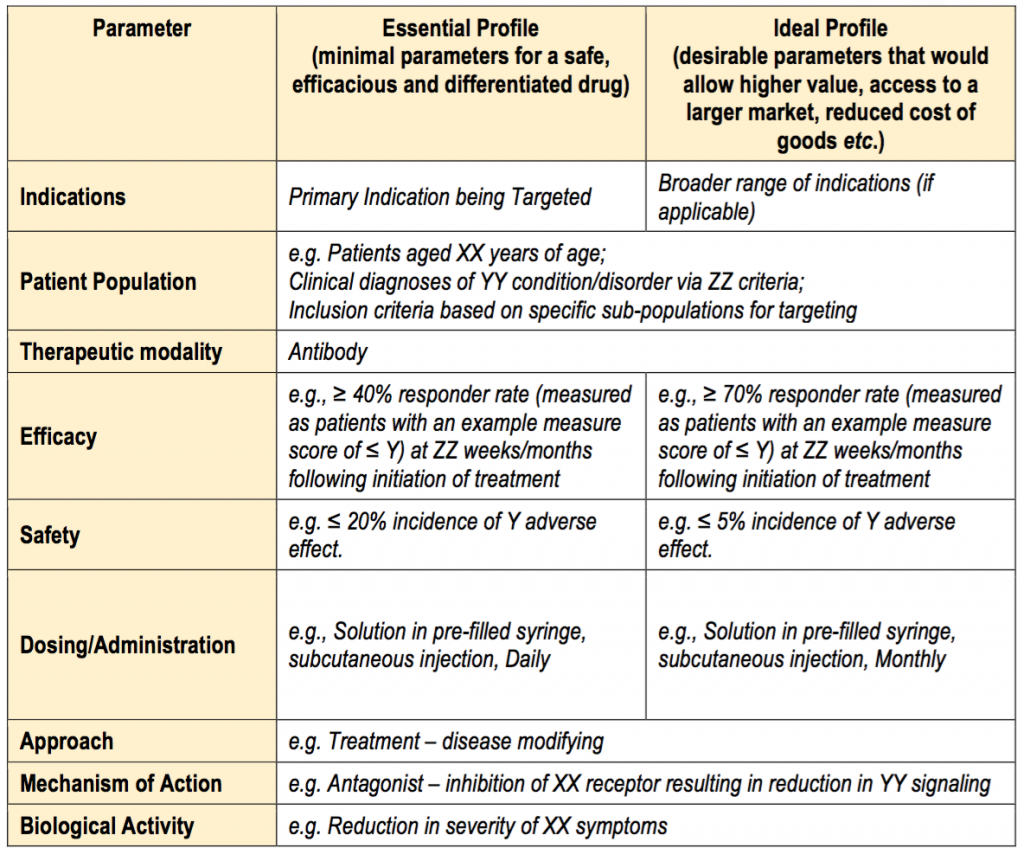
For more examples, visit the WHO target product profiles (TPPs) and product profile characteristics (PPCs) list and the NIH National Institute of Neurological Disorders and Stroke worksheets.
For further information on this, or any other topics related to the drug discovery and translation process, please email the BioCurate team on info@biocurate.com.

